The Genomics of COVID-19 Vaccine-Related Adverse Events project is one of the Global COVID Vaccine Safety (GCoVS) project* activities.
The GCoVS project affords a unique opportunity to examine genetic contribution towards vaccine-induced adverse events. Genetic contributions to serious and life-threatening drug reactions have seen genetic information incorporated into 800 drug labels worldwide by regulators. Genomic investigations are likely to inform who is at risk of a specific adverse event as well as lead to a better understanding of the biological or pathophysiological basis of adverse events.
The Genomics of COVID-19 Vaccine-Related Adverse Events project is being led by Dr Bruce Carleton and is focused on three adverse events that have been reported globally following COVID-19 vaccination, Guillain-Barré syndrome (GBS), vaccine-induced immune thrombotic thrombocytopenia (VITT)/ thrombosis with thrombocytopenia syndrome (TTS) and myocarditis and/or pericarditis. Participants are being sought from around the world and can self-refer.
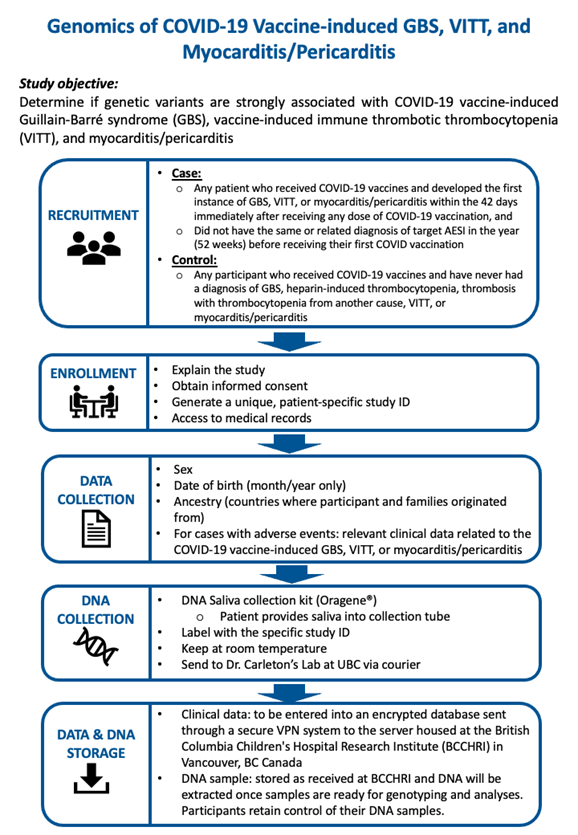
People interested in contributing a saliva sample for DNA analysis and/or researchers interested in collaborating in this study, please contact Dr Bruce Carleton by email for further information, bcarleton@popi.ubc.ca. An overview of the project methodology is shown in Figure 1.
Figure 1. Overview of genomics project methodology
Dr Carleton is the:
- Lead for the GVDN Global COVID Vaccine Safety (GCoVS) project genomics protocol work group
- Professor and Chair, Division of Translational Therapeutics, Department of Pediatrics, Faculty of Medicine, The University of British Columbia, Canada
- Senior Clinician Scientist, BC Children’s Hospital Research Institute, Canada
- Director, Pharmaceutical Outcomes Programme, BC Children’s Hospital, Canada
- Director, Therapeutic Evaluation Unit, Provincial Health Services Authority, Canada
*This project was supported by the Centers for Disease Control and Prevention (CDC) of the U.S. Department of Health and Human Services (HHS). The contents are those of the author and do not necessarily represent the official views of, nor an endorsement, by CDC/HHS, or the U.S. Government.
